~The objective to this project was to build a house that was adaptable to different weather conditions without using electricity.
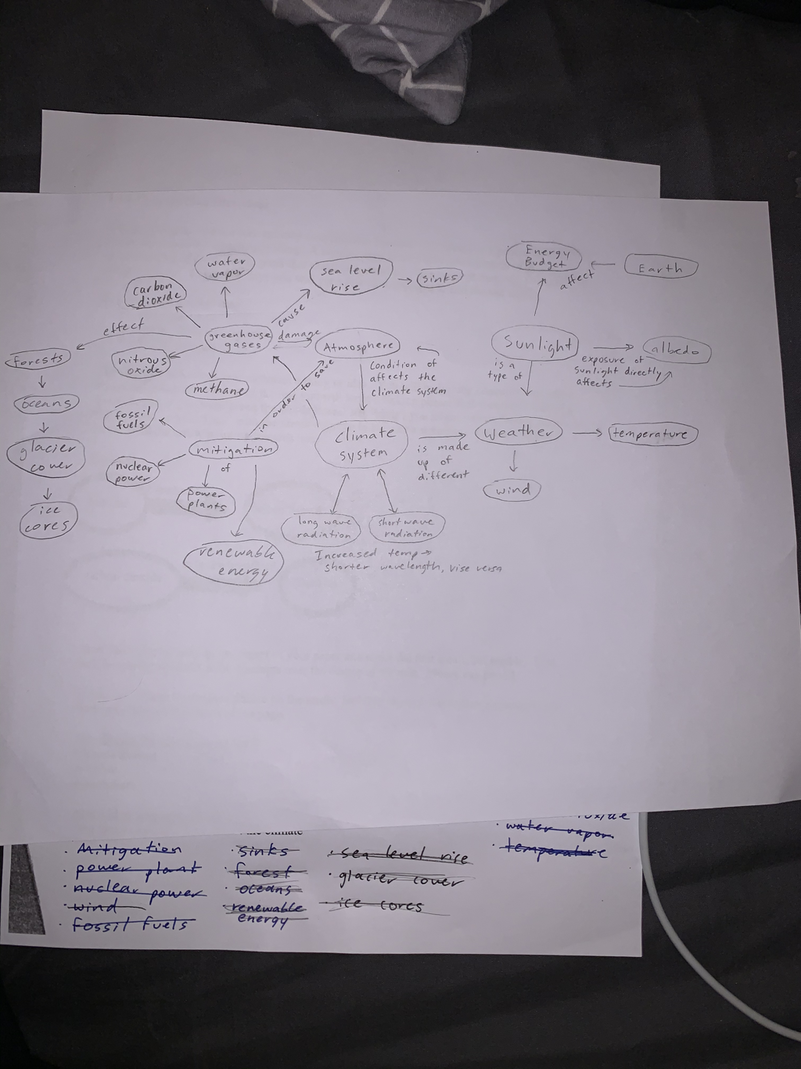
Heat (Q): a form of energy associated with the movement of atoms and molecules in any material. The higher the temperature of a material, the faster the atoms are moving, which means there will be a greater amount of energy present as heat. Q = mcΔT
Specific Heat Capacity (c): An object that cannot hold heat well meaning that the object heats up and cools down quickly. As an object with high specific heat capacity can hold higher temperature well, thus the object heats and cools slowly. It is the heat capacity per unit mass of a material.
Heat Transfer:
Radiation: The transfer of heat through any medium
Convection: The transfer of heat through liquids
Conduction: The transfer of heat through solids
Daylighting:
Solar Tubes:Sometimes called “light tubes,” and even “light tunnels,” have become the ideal solution for lighting interiors in a cost-effective, energy-efficient and eco-friendly way because they significantly reduce the need for electricity while keeping people connected to the outdoor environment.
Sky Lights: a window placed in a roof or ceiling to provide light
Clerestory Windows: a large window or many small windows on a high wall of a building. In a large building, the windows will be large to bring light into a large interior.
Light shelves: a horizontal surface that reflects daylight deep into a building. Light shelves are placed above eye-level and have high-reflectance upper surfaces, which reflect daylight onto the ceiling and deeper into the space.
Window Orientation: The windows should be south-westerly, as the sun is at a lower angle during the winter, if you also want to maximize solar time even during the evenings, rays of the sun are going towards North, so placing windows to the south-side allows for more rays to hit the house. Since the sun also sets west, westerly windowsill help capture some light before sunset.
Heat (Q): a form of energy associated with the movement of atoms and molecules in any material. The higher the temperature of a material, the faster the atoms are moving, which means there will be a greater amount of energy present as heat. Q = mcΔT
Specific Heat Capacity (c): An object that cannot hold heat well meaning that the object heats up and cools down quickly. As an object with high specific heat capacity can hold higher temperature well, thus the object heats and cools slowly. It is the heat capacity per unit mass of a material.
Heat Transfer:
Radiation: The transfer of heat through any medium
Convection: The transfer of heat through liquids
Conduction: The transfer of heat through solids
Daylighting:
Solar Tubes:Sometimes called “light tubes,” and even “light tunnels,” have become the ideal solution for lighting interiors in a cost-effective, energy-efficient and eco-friendly way because they significantly reduce the need for electricity while keeping people connected to the outdoor environment.
Sky Lights: a window placed in a roof or ceiling to provide light
Clerestory Windows: a large window or many small windows on a high wall of a building. In a large building, the windows will be large to bring light into a large interior.
Light shelves: a horizontal surface that reflects daylight deep into a building. Light shelves are placed above eye-level and have high-reflectance upper surfaces, which reflect daylight onto the ceiling and deeper into the space.
Window Orientation: The windows should be south-westerly, as the sun is at a lower angle during the winter, if you also want to maximize solar time even during the evenings, rays of the sun are going towards North, so placing windows to the south-side allows for more rays to hit the house. Since the sun also sets west, westerly windowsill help capture some light before sunset.
Solar Hot Water Heater
Project Objective: To build a heater to heat water as much as possible using solar.
Our group had many different ideas about what the final product of the solar heater may look like We pondered through many different ideas and finally decided that a simple design would work best for our group.
The contains a cardboard box, aluminum, to reflect the light, which carries heat, a water bottle, so that there is something to put the water in, and saran wrap, that would be used to trap the heat in the box. It was then set at a 60° angle, on the stand to get the rays of light coming in at 30° to get the best results.
Our device could heat up 350 mL of water, over a 70 minute period from 18°C to 40°C. The total heat gain in Joules was 32,223.2 J.
Project Objective: To build a heater to heat water as much as possible using solar.
Our group had many different ideas about what the final product of the solar heater may look like We pondered through many different ideas and finally decided that a simple design would work best for our group.
The contains a cardboard box, aluminum, to reflect the light, which carries heat, a water bottle, so that there is something to put the water in, and saran wrap, that would be used to trap the heat in the box. It was then set at a 60° angle, on the stand to get the rays of light coming in at 30° to get the best results.
Our device could heat up 350 mL of water, over a 70 minute period from 18°C to 40°C. The total heat gain in Joules was 32,223.2 J.
Day lighting Lab
Our group had tested solar tubes, skylights, light shelves, windows, reflective materials ect. We tested these against non reflective, dark, and light surfaces. Of course different shades reflect different amounts of light allowing more light. After testing we concluded that the southern light shelf was the most effective. Lighter colored interiors were also better for lighting, but performed the about the same as reflective aluminum.
Our group had tested solar tubes, skylights, light shelves, windows, reflective materials ect. We tested these against non reflective, dark, and light surfaces. Of course different shades reflect different amounts of light allowing more light. After testing we concluded that the southern light shelf was the most effective. Lighter colored interiors were also better for lighting, but performed the about the same as reflective aluminum.
Reflection
Materials heating lab
Analysis:
Over the course of this project we collected large amounts of data about different about different materials. We learned that if the temperature of a material rises quickly, it has a low heat capacity. If the temperature rises slowly, the material has a high heat capacity. The higher the heat capacity the hotter it can be without the material itself getting too hot.
During this lab we came across some errors. Some of them were different heights of the lamps, possible temperature differences, and possible timing errors. We hung the lights on cabinets above countertops where the object layed. The heights of the lamps may have been inconsistent because it was tough to hang them all at the same heights when you had to tie them to the handles of the cabinets and hang them from the edge of the cabinet door. The next flaw was use of different heat lamps that may of had different temperature outputs. There were different types of heat lamps that were used so there was a possibility that one was hotter than the other or one was not able to heat up to the level of the other one. The last error was possible errors in the timing of how long the object was exposed to the heat lamp. We had designated timers and there was a possibility they could have made errors.
There were many ways we could improve on this experiment and fix these errors. One way would be to use longer time heating intervals. We only got to test the materials for 10 minutes at a time so if we could test them for longer than we could get a better idea on how they actually react to heat. Another way to improve our project was to keep track of not only the heating time but also the cooling time of the material. It is important to know how long the material will keep the heat in the house because if the heat escapes quickly then it would be cold in the later hours of the night when there is no sunlight.
This experiment may lead into some further experiments. One of them may be the idea of waterproofing of the materials. If the roof can't hold out water then in the long run it doesn't really matter how well it can hold heat in. Another experiment this specific experiment may lead into is the stability check of how well these different materials hold up to various outside forces. These materials would be exposed to many different conditions. We would have to test how well they held up, if they collapsed, corroded, or crumbled under the pressure of these conditions.
Over the course of this lab we learned many things. One of the most important ideas was that the density of the materials made a difference in the temperature of them. The objects with a higher density heat up slower because there is more mass to cover while with a smaller less dense object heat up quickly because there is less mass to heat up. This was represented many times in our lab. One time is was shown was the between the brick and the carpet. The brick of course being the more dense object and the carpet being the less dense. The brick started at 22 degrees and increased to 36.5 degrees. The carpet however started at 23 degrees and increased to 53 degrees. This proves the theory that the smaller less dense objects did heat up faster and the larger more dense objects heat up slower. This is only one of the examples of this theory in this lab with many more to prove this argument.
Reflection
Materials heating lab
Analysis:
Over the course of this project we collected large amounts of data about different about different materials. We learned that if the temperature of a material rises quickly, it has a low heat capacity. If the temperature rises slowly, the material has a high heat capacity. The higher the heat capacity the hotter it can be without the material itself getting too hot.
During this lab we came across some errors. Some of them were different heights of the lamps, possible temperature differences, and possible timing errors. We hung the lights on cabinets above countertops where the object layed. The heights of the lamps may have been inconsistent because it was tough to hang them all at the same heights when you had to tie them to the handles of the cabinets and hang them from the edge of the cabinet door. The next flaw was use of different heat lamps that may of had different temperature outputs. There were different types of heat lamps that were used so there was a possibility that one was hotter than the other or one was not able to heat up to the level of the other one. The last error was possible errors in the timing of how long the object was exposed to the heat lamp. We had designated timers and there was a possibility they could have made errors.
There were many ways we could improve on this experiment and fix these errors. One way would be to use longer time heating intervals. We only got to test the materials for 10 minutes at a time so if we could test them for longer than we could get a better idea on how they actually react to heat. Another way to improve our project was to keep track of not only the heating time but also the cooling time of the material. It is important to know how long the material will keep the heat in the house because if the heat escapes quickly then it would be cold in the later hours of the night when there is no sunlight.
This experiment may lead into some further experiments. One of them may be the idea of waterproofing of the materials. If the roof can't hold out water then in the long run it doesn't really matter how well it can hold heat in. Another experiment this specific experiment may lead into is the stability check of how well these different materials hold up to various outside forces. These materials would be exposed to many different conditions. We would have to test how well they held up, if they collapsed, corroded, or crumbled under the pressure of these conditions.
Over the course of this lab we learned many things. One of the most important ideas was that the density of the materials made a difference in the temperature of them. The objects with a higher density heat up slower because there is more mass to cover while with a smaller less dense object heat up quickly because there is less mass to heat up. This was represented many times in our lab. One time is was shown was the between the brick and the carpet. The brick of course being the more dense object and the carpet being the less dense. The brick started at 22 degrees and increased to 36.5 degrees. The carpet however started at 23 degrees and increased to 53 degrees. This proves the theory that the smaller less dense objects did heat up faster and the larger more dense objects heat up slower. This is only one of the examples of this theory in this lab with many more to prove this argument.
Reflection
Overall, I believe this very long project went pretty well for us. We had a leader being Nihal, a task manager being myself, a helper Thomas, and a big thinker and task completer being Henry. We did our work quite smoothly, and we rarely ran into problems. Our main peaks were assigning roles and collaborating well. When we were assigning roles, we had no problems. Everybody gladly excepted their roles, and there was no arguing about it. The main pit in our project was getting our work done early or even on time. We struggled finishing the presentation. In the future we all need to work on managing our time wisely and being productive with every minute there is to work.