Introduction
For this project our classes' task was to create a board game that incorporated different chemicals and their reactions. This was a fun way of going about that because it made it interactive and easier to learn. Our main topics for this project were reading and writing chemical equations, single replacement reactions, double displacement reactions, creation of a gas, and dealing with LEDs and Breadboards. Keeping that in mind my group decided to create a board game that was based off of the game candy land. Our game consisted of a series of chemical reactions while playing the board game in a very similar manner to candy land. You would chose a game piece and select a card with a color on it corresponding to different spaces on the board. Once certain spaces on the board were reached the participant would perform a chemical reaction in order to move on to the next space.
Instructions
For our project we types up an instruction manual that included the reactions that took place during game play. In addition to the instructions and chemical reactions listed, we also included safety precautions for our game.
For this project our classes' task was to create a board game that incorporated different chemicals and their reactions. This was a fun way of going about that because it made it interactive and easier to learn. Our main topics for this project were reading and writing chemical equations, single replacement reactions, double displacement reactions, creation of a gas, and dealing with LEDs and Breadboards. Keeping that in mind my group decided to create a board game that was based off of the game candy land. Our game consisted of a series of chemical reactions while playing the board game in a very similar manner to candy land. You would chose a game piece and select a card with a color on it corresponding to different spaces on the board. Once certain spaces on the board were reached the participant would perform a chemical reaction in order to move on to the next space.
Instructions
For our project we types up an instruction manual that included the reactions that took place during game play. In addition to the instructions and chemical reactions listed, we also included safety precautions for our game.
Proof of project
Images of our project completed
Images of our project completed
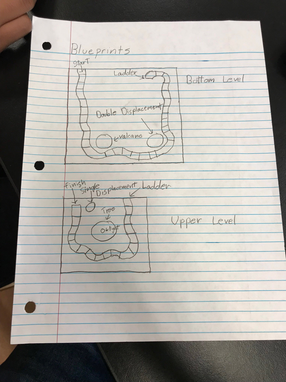
Blueprints
Along with actually creating this board game, we had to draw up blueprints that showed our project from a different view. It showed all of the details of our project and where the reactions were placed along the board.
Along with actually creating this board game, we had to draw up blueprints that showed our project from a different view. It showed all of the details of our project and where the reactions were placed along the board.
Chemicals and Their Reactions
Single replacement reaction: A single replacement reaction is a type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound.
In our case we used copper nitrate and aluminum foil. These two reacted in a way to where the copper nitrate disintegrated the aluminum. Aluminum (Al(s)) and copper nitrate (Cu(NO3)(l)) react to form copper (Cu(s)) and aluminum nitrate (Al(NO3)3.
In our project we used the single replacement reaction at the end of our game. The purpose was for the tin foil to break apart lighting up the LED on the breadboard which symbolized a star on the top of a Christmas tree meaning the game was over.
Double replacement reaction: A double displacement reaction, also known as a double replacement reaction or metathesis, is a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products.
In our case we used potassium iodide and lead nitrate and mixed those two to create a chemical reaction. These two chemicals reacted in a way that created a yellow substance that we designated to be a lemon flavored candy. Once the player reached the space correlating with this reaction they would pour the two chemicals into a vile and water down the solution to make the chemical reaction happen. Lead Nitrate, Pb(NO3)2(aq), and Potassium Iodide, KI(aq), to form the insoluble salt Lead Iodide, PbI2(s); which precipitates out as a bright yellow solid. At the same time, the water soluble salt Potassium Nitrate, KNO3(aq), is also formed.
Production of a gas: a mixture of two substances that emits a non harmful gas.
In our case we mixed baking soda and vinegar to create a small "explosion" of a volcano. The mixture of baking soda and vinegar react to create a fizzy solution that spews out of the volcano. Once the participant reached the designated area for the reaction they would pour 45 ml of vinegar into the volcano and add a spoonful of baking soda to the liquid causing a reaction that releases carbon dioxide. Baking soda and vinegar react with each other because of an acid-base reaction. Baking soda is a bicarbonate (NaHCO3) and vinegar is an acetic acid (HCH3COO). This tied into our project being a Christmas volcano and exploded to signify you were doing a good job, a congratulatory explosion of sorts.
Single replacement reaction: A single replacement reaction is a type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound.
In our case we used copper nitrate and aluminum foil. These two reacted in a way to where the copper nitrate disintegrated the aluminum. Aluminum (Al(s)) and copper nitrate (Cu(NO3)(l)) react to form copper (Cu(s)) and aluminum nitrate (Al(NO3)3.
In our project we used the single replacement reaction at the end of our game. The purpose was for the tin foil to break apart lighting up the LED on the breadboard which symbolized a star on the top of a Christmas tree meaning the game was over.
Double replacement reaction: A double displacement reaction, also known as a double replacement reaction or metathesis, is a type of chemical reaction where two compounds react, and the positive ions (cation) and the negative ions (anion) of the two reactants switch places, forming two new compounds or products.
In our case we used potassium iodide and lead nitrate and mixed those two to create a chemical reaction. These two chemicals reacted in a way that created a yellow substance that we designated to be a lemon flavored candy. Once the player reached the space correlating with this reaction they would pour the two chemicals into a vile and water down the solution to make the chemical reaction happen. Lead Nitrate, Pb(NO3)2(aq), and Potassium Iodide, KI(aq), to form the insoluble salt Lead Iodide, PbI2(s); which precipitates out as a bright yellow solid. At the same time, the water soluble salt Potassium Nitrate, KNO3(aq), is also formed.
Production of a gas: a mixture of two substances that emits a non harmful gas.
In our case we mixed baking soda and vinegar to create a small "explosion" of a volcano. The mixture of baking soda and vinegar react to create a fizzy solution that spews out of the volcano. Once the participant reached the designated area for the reaction they would pour 45 ml of vinegar into the volcano and add a spoonful of baking soda to the liquid causing a reaction that releases carbon dioxide. Baking soda and vinegar react with each other because of an acid-base reaction. Baking soda is a bicarbonate (NaHCO3) and vinegar is an acetic acid (HCH3COO). This tied into our project being a Christmas volcano and exploded to signify you were doing a good job, a congratulatory explosion of sorts.
Reflection
Overall I felt this project went very well. Throughout the building process and creation of the chemical reactions we all performed well and worked hard to create a game we were all satisfied with. As for me I felt I was less of a facilitator but when I was asked to perform a task I always carried through with it without an argument. I felt I was one of the most productive members when it came to the actual hands on building of the board.
There were many things that our group did well together and some we didn't. One of the aspects I feel we excelled in was collaboration. Luckily I knew the people who were in my group so collaborating with them and working together with them was fairly effortless. We could all assign each other tasks without having to doing it forcefully because of how well we worked with each other. Another thing I felt we did well was work efficiently. For the majority of this project we were working hard and not messing around. We knew the deadline for the final product and worked hard accordingly. With one of our group members leaving toward the beginning of the project it put more of a work on the three remaining including me. Even with this setback we were still able to complete all of our work in a timely manner
Along with the good things There were also some things we may have done poorly. One of these could have been me personally being off task at times during the project. At times where I thought there may not have been much to work on I would wander to other groups. This wasn't fair to my other group members and will not happen in future projects. Another thing that I may have done poorly was fully contribute to the document we created for the project. Although I contributed well during the building process I felt I could have done a better job when it came to the electronical part of the project.
Summary
Overall this was a very enjoyable project that taught me a lot about chemical reactions and electrical currents involving breadboards. I would recommend this to students and teachers because it was a very fun, interactive way to experience and learn about these reactions.
Overall I felt this project went very well. Throughout the building process and creation of the chemical reactions we all performed well and worked hard to create a game we were all satisfied with. As for me I felt I was less of a facilitator but when I was asked to perform a task I always carried through with it without an argument. I felt I was one of the most productive members when it came to the actual hands on building of the board.
There were many things that our group did well together and some we didn't. One of the aspects I feel we excelled in was collaboration. Luckily I knew the people who were in my group so collaborating with them and working together with them was fairly effortless. We could all assign each other tasks without having to doing it forcefully because of how well we worked with each other. Another thing I felt we did well was work efficiently. For the majority of this project we were working hard and not messing around. We knew the deadline for the final product and worked hard accordingly. With one of our group members leaving toward the beginning of the project it put more of a work on the three remaining including me. Even with this setback we were still able to complete all of our work in a timely manner
Along with the good things There were also some things we may have done poorly. One of these could have been me personally being off task at times during the project. At times where I thought there may not have been much to work on I would wander to other groups. This wasn't fair to my other group members and will not happen in future projects. Another thing that I may have done poorly was fully contribute to the document we created for the project. Although I contributed well during the building process I felt I could have done a better job when it came to the electronical part of the project.
Summary
Overall this was a very enjoyable project that taught me a lot about chemical reactions and electrical currents involving breadboards. I would recommend this to students and teachers because it was a very fun, interactive way to experience and learn about these reactions.